Table of Contents
Over the years, antibiotics have been used in food animal production to treat and prevent disease. Due to penicillin’s widespread manufacture at the time to treat war casualties, veterinarians were able to access lyophilized versions of the drug during the closing stages of World War II, which led to the first use of antibiotics in food animals. Penicillin has been reported to be more effective in treating some ailments than the previously available medications since it is an antibiotic.
Importance of Antibiotics
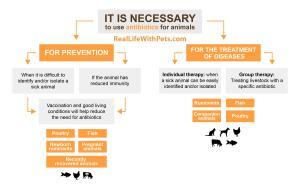
Antibiotics are essential for fighting animal disease. There is simply no other way to treat a bacterial infection. Antibiotics treat painful illnesses in suffering animals and halt the spread of infectious diseases. Therefore, healthy animals can produce milk, meat, and eggs that are safe for human consumption and free of harmful bacteria. ‘Withdrawal Periods’ guard against harmful trace amounts or ‘antibiotic residues’ in these foods.
These medicines are among the most valuable parts of a veterinarian’s kit and directly support animal health and welfare. However, antibiotic resistance can put their effectiveness at risk, threatening our ability to respond to deadly bacterial diseases.
Animals are vulnerable to some of the same bacterial infections as people, such as pneumonia and skin infections that can be treated with antibiotics. These medicines treat not only the disease but also the animal, reducing the length of sickness and the animal’s suffering and contributing to their welfare and well-being.
Livestock producers lose countless animals each year due to diseases. That’s why the ability to keep animals healthy is what allows them to provide for their family. For a developing world farmer, livestock is a form of savings and investment. However, losing a single animal can put their entire livelihood, as well as food security, in jeopardy.
Prevention Measures
Humans must use prevention strategies including vaccination, biosecurity, diagnostics, and nutrition as their first line of defense against life-threatening illnesses. However, antibiotics continue to be the only option for treating a bacterial infection when sickness bypasses these defenses.
Animals need less food and water when they are healthy because they can process their food and water more efficiently. Additionally, more productive animals produce more and higher quality meat, milk, and eggs.
On the other hand, sick animals have a dramatic increase in emissions. For example, a sickness in the cattle can raise GHG emissions by up to 24% of every unit of milk and 113% per beef carcass.
What’s more, radically slashing antibiotic use – such as in a ‘Raised Without Antibiotics’ system — can hurt animal welfare, according to the American Veterinary Medical Association (AVMA).
Abandoning antibiotics in the U.S. could also mean more than 680 million additional broiler chickens would need to be raised annually to meet demand. Such an increase would also bring added environmental pressures, needing more than 1.9 billion extra gallons of water and more than 5.4 million additional tons of feed per year.
Ultimately, responsible antibiotic use can support good animal welfare, health, and sustainability, combined with other tools like proper vaccination, biosecurity, good nutrition, rapid diagnostics, and more.
Early Diagnosis
Animal illness detection is a difficult task. Animals cannot tell us their symptoms or let us know when they are getting sick. Instead, vets rely on additional indicators such as changes in behavior or physical symptoms, reports of disease in the neighborhood, and knowledge gained from earlier epidemics. However, an animal may be ill long before any symptoms show up or a clinical diagnosis may be made.
Therefore, a veterinarian will treat an animal with an antibiotic when they determine it has a bacterial infection and determine the likelihood that the infection will not spread to other animals on the farm or elsewhere.
The veterinarian may discover that other animals are significantly in danger, likely to get sick, or may already be unwell but not yet showing signs using knowledge of how illnesses spread. When this is the case, they could suggest an antibiotics course to help the animals’ infection be controlled.
Antibiotic treatment for a herd or flock stops a disease threat in the bud rather than allowing it to spread and cause suffering before signs become visible or animals start to die.
‘Raised Without Antibiotics’ System’s Fail
Imagine being a veterinarian who finds a sick pig on a farm and diagnoses it with a bacterial infection before prescribing an antibiotic course. However, you are also aware that the infection is contagious before any noticeable symptoms show up or a clinical diagnosis can be made. Therefore, even if they are not yet showing symptoms, you might presume that many other animals are already ill.
As the vet, you may either wait for the other animals to start showing symptoms, or you can recommend an antibiotic treatment to control the infection and prevent it from spreading into a more severe outbreak.
Although every case is different, veterinarians must determine the risk to other animals of allowing infected animals to stay untreated. However, suppose they are aware that an animal is likely already ill. In that case, they understand that it is better, kinder, and more cost-effective to stop the threat before it becomes apparent to the animal that it is suffering.
There are many factors that can prevent sicknesses, such as excellent nutrition, exercise, and hygiene, but when bacterial disease occurs, there is no cure other than antibiotics. Animals who are ill and are not given the proper medication will experience severe symptoms or perhaps pass away.
Producers are shifting to “Raised Without Antibiotics” or “No Antibiotics Ever” systems in some countries. Because they think these solutions are better for animal welfare, besides consumers push for them. A recent poll of farmers found that they believed “antibiotic-free” farming methods were more harmful to animal welfare. Those who have worked on “antibiotic-free” farms observed increased disease rates and said that, occasionally, keeping the “antibiotic-free” label took precedence over the well-being of the animals.
According to estimates from the Chief Economist of the American Veterinary Medical Association (AVMA), hens that are “raised without antibiotics” have a death rate that is 25–50% higher.
Without antibiotics, treating an animal for a bacterial infection is impossible. By preventing and controlling disease, we must put human attention on lowering the demand for antibiotics.
Are Antibiotics in Our Food?
Animal antibiotic use is strictly regulated to ensure there are no dangerous “residues” (i.e., trace amounts) in our food. To prevent milk, meat, and eggs from entering the food chain before an animal has metabolized an antibiotic enough, strict “withdrawal periods” should be followed.
The research and control of antibiotics are based on food safety. Therefore, every antibiotic has a distinct “withdrawal time” that specifies how many days must pass after antibiotic treatment for a livestock farmer to allow their animal or its produce to be consumed.
This withdrawal period allows time for the animal to sufficiently processed the antibiotic. To ensure that users are aware of their responsibilities, they are printed on the antibiotic label. It guarantees that hazardous “residues,” or trace amounts of a drug, are not present in milk, meat, or eggs.
International laws governing withdrawal times and “maximum residue limits” are established and upheld, and both are reviewed as part of the approval process for medicines. To ensure that withdrawal times are followed, governments also randomly test farm-produced food for dangerous antibiotic residues. Violations may result in severe penalties.
Does Antibiotic Use in Animals Lead to Resistant Bacteria in People?
People’s overuse of antibiotics is the leading cause of antibiotic resistance in humans. However, the appropriate use of antibiotics in animals can reduce resistance.
Antibiotic resistance is a complex global health threat. Although scientists currently have a limited knowledge of how and how frequently this occurs, bacteria — whether resistant or not — can spread from humans to animals.
The use of antibiotics in animals plays a role in the development of resistance. Still, the European Centre for Disease Prevention and Control claims that the use of antibiotics in human medicine continues to be the predominant factor in the development of resistance in humans.
That is why other research shows that if action is only taken in animals, AMR in people will likely remain unchecked.
Additionally, according to the OECD, three out of four deaths may be prevented by spending just $2 per person a year on measures as simple as handwashing and more sensible antibiotic prescription.
More research is required to completely comprehend the connection between drug-resistant microorganisms in people and animals. However, responsible use and actions with observable effects must be prioritized in the meantime by both sides.
A vital element is reducing the need for these medicines rather than simply reducing their use. Therefore, making the most of all measures, including vaccination, effective biosecurity, husbandry procedures, and proper nutrition, that contribute to illness prevention in the first place is necessary.
What Is Ashoxy?
This medicine is widely used in the modern veterinary field to treat many animal diseases. Ashoxy is a drug from the group of antibiotics, and its composition is balanced and optimal for treating a whole range of diseases. This remedy was invented in India, and today it is known in many countries.
to treat many animal diseases. Ashoxy is a drug from the group of antibiotics, and its composition is balanced and optimal for treating a whole range of diseases. This remedy was invented in India, and today it is known in many countries.
Ashoxy was created by Ashish Life Sciences Pvt. Ltd. This company has a high reputation in the market of pharmacological products in Asia and worldwide. Ashish Life Sciences Pvt. Ltd. was founded in 1974 in Mumbai, Maharashtra, India. This brand is India’s leading exporter and supplier of all veterinary medicine. With ext
ensive experience in the supply and trade of Ashoxy, Ashish Life Sciences Pvt. Ltd. has made a well-known n
ame for itself in the market.
The medication comes in the form of a spray and tablets. The product has been tested several times before launching to the pharmaceutical market, and its effectiveness is confirmed. Ashoxy is designed with the utmost preci
sion. It consists of high-quality ingredients. Thanks to advanced technologies, the product turned out to be effective and safe.
Ashoxy is produced in 2 forms – Ashoxy Spray, 200ml, and Ashoxy 1000 Tablets; each tablet contains 1000 mg of oxytetracycline hydrochloride with 50 or 100 tablets in the pack.
Oxytetracycline is an active ingredient of Ashoxy. Oxytetracycline is a broad spectrum antibiotic active against many Gram-positive and Gram-negative bacteria, certain mycoplasma species, chlamydia, rickettsia (e.g., Rocky Mountain spotted fever), and some protozoa. In addition, it exerts its antibacterial action by inhibiting the protein synthesis of organisms.
Oxytetracycline is a broad-spectrum tetracycline antibiotic, the second of the group to be discovered. Other related drugs in this class include doxycycline, tetracycline, and minocycline. It is effective against bacteria that infect the eyes, oral cavity, bone, respiratory tract, sinuses, and blood cells.
| Oxytetracycline Physicochemical Properties | |
| Parameter | Value |
| Boiling point (°C) | not available |
| Melting point (°C) | 179-182 (decomoses) |
| pH | 6.115 (saturated solution) |
| Density (kg/m3) | not known |
| Flash point (°C) | not available |
| Stability | Stable in air, light sensitive |
| Solubility | Very slightly soluble in water; freely soluble in 3 NHCL and in alkaline solutions; sparingly soluble in alcohol |
| Solubility – monoalkyl (c8-C18) trimethyl ammonium salt | 1.0 mg/ml in water |
| Solubility-dihydrate | 0.6 mg/ml in water |
History of Oxytetracycline
After introducing chlortetracycline (Aureomycin) in 1948, this antibiotic was rapidly recognized as an essential and highly effective agent against a broad spectrum of infectious diseases. Oxytetracycline, introduced two years later, was soon found to have an almost identical spectrum of activity. As a result, these antibiotics are now undisputedly ranked among the most important antimicrobial agents available.
The antibiotic was patented in 1949 and came into commercial use in 1950. It is on the World Health Organization’s List of Essential Medicines. It was first found near Pfizer laboratories in a soil sample yielding the soil actinomycete, Streptomyces rimosus by Finlay et al.
In 1950, a group at Pfizer led by Francis A. Hochstein, working in a loose collaboration with the Harvard organic chemist, worked out the chemical structure of oxytetracycline, enabling Pfizer to mass-produce the drug under the trade name Terramycin. This discovery was a significant advancement in tetracycline research. In addition, it paved the way for discovering an oxytetracycline derivative, doxycycline, one of the most popularly used antibiotics today.
Oxytetracycline is used to control the outbreak of American foulbrood and European foulbrood in honeybees.
American livestock producers apply oxytetracycline to livestock feed to prevent diseases and infections in cattle and poultry.
It is important to note that Ashoxy is for veterinary purposes only. While there are types available for human use, you should only use the kind prescribed by a veterinary doctor or animal care specialist for an animal.
Clinical Pharmacology
Oxytetracycline is an antibiotic that belongs to the group of tetracyclines.
The mode of action is inhibition of protein synthesis by binding to 30S ribosomal subunit of microorganisms susceptible to oxytetracycline, thereby preventing the connection between ribosomes and aminoacyl-tRNA.
It’s also believed that oxytetracycline inversely connects with 50S ribosomes and changes the permeability of the cytoplasmic membrane of the sensitive microorganisms. Oxytetracycline, at high concentrations, can also inhibit protein synthesis in cells.
Oxytetracycline penetrates practically into all organs and tissues, but the highest level occurs in the lungs, liver, bile, reproductive organs, and kidneys. At low concentrations, oxytetracycline appears in the cerebrospinal fluid, but its concentration does not reach therapeutic levels. Oxytetracycline does not form metabolites. It is excreted with urine and bile as well as with milk.
Oxytetracycline belongs to a structurally diverse class of aromatic polyketide antibiotics produced by Streptomyces via type II polyketide synthases (PKSs), also known as bacterial aromatic polyketides. Other compounds produced via type II PKSs are essential bioactive compounds that span from anticancer agents doxorubicin to antibiotics such as tetracycline.
The biosynthesis of oxytetracycline can be broken down into three general portions:
- The formation of an amidated polyketide backbone with minimal PKS’s.
- The cyclization of the polyketide backbone.
- The formation of anhydrotetracycline, a shared intermediate with tetracycline, to produce oxytetracycline.
The biosynthesis of oxytetracycline begins with the utilization of PKS enzymes ketosynthase (KS), the chain length factor (CLF), the acyl carrier protein (ACP), and an acyltransferase (encoded as OxyA, OxyB, OxyC and OxyP in the oxytetracycline gene cluster) to catalyze the extension of the malonyl-CoA starting unit with eight malonyl-CoA extender units. Then, the process of elongating the polypeptide skeleton occurs through a series of Claisen-like decarboxylation reactions until the linear tetracyclic skeleton is formed. Thus, minimal PKS’s form a completed amidated polyketide backbone without any additional post-synthase tailoring enzymes.
Effectiveness
Oxytetracycline works by interfering with the ability of bacteria to produce essential proteins. Without these proteins, the bacteria cannot grow, multiply and increase in numbers. Oxytetracycline stops the spread of the infection, and the remaining bacteria are killed by the immune system or eventually die.
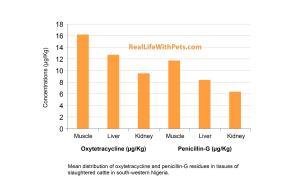
Oxytetracycline is more effective than liquamicyn, providing the immediate impact and long-lasting treatment animals need to recover.
Compared to those treated with liquamicyn, cattle treated with oxytetracycline had higher peaks and longer duration of antibiotic levels in tissue.
| Effect of Treatments with Oxytetracycline on Dogs Naturally Infected with Rickettsiales | |||
| Animal | Before Treatments (Day 0) | During Treatments (Day 4) | After Treatments (Day 8) |
| 1 | Positive | Positive | Negative |
| 2 | Positive | Positive | Negative |
| 3 | Positive | Negative | Negative |
| 4 | Positive | Negative | Negative |
| 5 | Positive | Negative | Negative |
Composition of Ashoxy
The main ingredient in the composition is oxytetracycline hydrochloride. This well-known antibiotic has a wide range of effects. Its action has a solid antibacterial nature, due to which protein synthesis is suppressed.
The Ashoxy Spray comes in a 200 ml volume. Each ml contains:
- Oxytetracycline Hydrochloride I.P.50 mg.
The Ashoxy 1000 Tablets come in 50 or 100 tablets in a pack. Each tablet contains:
- 1000 mg Ashoxy 1000 (Oxytetracycline) Hydrochloride.
Uses of Ashoxy
Ashoxy is indicated for treating diseases associated with oxytetracycline susceptible organisms like Bordetella, Campylobacter, Chlamydia, E. coli, Haemophilus, Mycoplasma, Pasteurella, Rickettsia, Salmonella, Staphylococcus, and Streptococcus spp. in cattle, goats, sheep, pig, and horses.
Ashoxy Spray is also designed for the topical treatment and prevention of skin diseases caused by bacteria sensitive to oxytetracycline in cattle, sheep, goats, pigs, horses, dogs, cats, rabbits, etc.
Ashoxy Tablets should be taken orally to treat wounds of traumatic origin; surgical wounds (castration, cesarean section, dehydration, cupping, ears, etc.); abrasions, scratches, puncture wounds; udder injuries during machine milking; necrobacteriosis of cattle and sheep; bacterial infections accompanying parasitic dermatitis; skin diseases due to its activity against the bacteria on the skin that influence the development of acne (Cutibacterium acnes), wound infections and acute metritis caused by strains of Staphylococci and Streptococci organisms sensitive to oxytetracycline.
Oxytetracycline treats respiratory tract infections (pneumonia), urinary tract, soft tissues, and skin. It is used for infections caused by a broad spectrum of bacteria, except that resistance is common among gram-negative bacilli of enteric origin and staphylococci. One of the most common uses is in cattle for treating bovine respiratory disease caused by Pasteurella multocida, Mannheimia haemolytica, and Histophilus somni.
In cattle, oxytetracycline is indicated in the treatment of the following conditions:
- pneumonia and shipping fever complex associated with Pasteurella and Haemophilus;
- infectious bovine kerato-conjunctivitis (pinkeye) caused by Moraxella Bovis;
- foot-rot and diphtheria caused by Fusobacterium necrophorum;
- bacterial enteritis (scours) caused by Escherichia coli;
- leptospirosis caused by Leptospira pomona;
- wooden tongue caused by Actinobacillus lignieresii;
- wound infections and acute metritis caused by strains of staphylococci and streptococci organisms sensitive to oxytetracycline.
In pigs, tetracyclines have been used to treat atrophic rhinitis, pneumonic pasteurellosis, Mycoplasma infections, and chronic bronchitis, due to its activity against the bacteria usually responsible, Haemophilus influenza; bacterial enteritis caused by Escherichia coli, pneumonia caused by Pasteurella multocida and leptospirosis caused by Leptospira pomona.
In sows, the medicine is indicated to aid in controlling infectious enteritis in suckling pigs caused by Escherichia coli.
In small animals, it is used as a treatment for rickettsia spp. and Ehrlichia spp. infections.
In horses, oxytetracycline has been administered to treat equine piroplasmosis caused by Theileria equi but was not effective for Babesia caballi. In addition, oxytetracycline has been used for treating Potomac fever in horses caused by Neorickettsia risticii, as well as respiratory and soft tissue infections. In newborn horses, oxytetracycline has been administered at high doses to correct angular limb deformities. The dosages have been as high as 50–70 mg/kg IV q48h. This effect may be caused by a decrease in the viscoelastic properties in the tendons of young animals. Because this produces tendon or ligament laxity, it may correct angular limb deformities in young foals.
In dogs and cats, oxytetracycline suppresses protein synthesis and growth, which prevents bacteria from growing. Although this drug is not approved for use in animals other than livestock by the Food and Drug Administration, it is prescribed legally by veterinarians as an extra-label drug.
Oxytetracycline is used to mark fish that are released and later recaptured. The oxytetracycline interferes with bone deposition, leaving a visible mark on growing bones.
Oxytetracycline has also been formulated as a broad-spectrum anti-infective for fish under the name Terramycin 200. It controls certain diseases that adversely affect salmonids, catfish, and lobsters.
What are the symptoms for prescribing Ashoxy?
These can be the following:
- various infections, including weakened immunity;
- general weakness of the animal;
- loss of weight and appetite;
- disorders of the digestive system;
- other problems with internal organs.
Only an experienced veterinarian can prescribe the drug, so consult your veterinarian before administering Ashoxy. He makes a precise diagnosis, and only after that the drug Ashoxy can be prescribed.
Read and follow all label directions.
Dosage Sizes
Before applying Ashoxy, it is recommended to clean the treated surface of pus, exudate from wounds, necrotized tissues, remove wool, etc.
Before using the spray, you need to shake well and spray the drug from a distance of 18-20 cm from the affected area for 2-3 seconds. The barrel is designed so that the medication can be sprayed even in the inverted position of the barrel. Ashoxy medication is recommended for the one-time treatment of the affected area. Repeated application of the drug is recommended no earlier than a week.
The amount of medication sprayed is determined based on the affected area. Combining topical treatment with complex therapy is recommended in cases of deep and complicated infections.
If necessary, repeated treatment is carried out every seven days until complete healing, but not more than four times.
Ashoxy Tablets should be taken orally by mixing into feed or drinking water.
Dosage for Ashoxy Tablets:
- Large Animals: 5 to10 mg/kg body weight repeated every 24 hours for 3-5 days.
- Small animals: 10mg/kg body weight 24 hours for 3-5 days.
The duration of administration depends on the condition being treated, response to the medication, and the development of any adverse effects. Usually, the duration of use of the Ashoxy is from 1 to 28 days. Be sure to complete the prescription unless specifically directed by your veterinarian. Even if your pet feels better, the entire treatment plan should be completed to prevent relapse or the development of resistance.
Alternatives should be sought if no improvement occurs within three months.
In the case of missing a dose, the course of treatment should be resumed at the doses and the scheme of use provided by your vet.
Withdrawal Time
The antibiotic is partially absorbed in the animal’s gastrointestinal tract, and the remaining is deposited in manure. Researchers at the Agricultural Research Service studied the breakdown of oxytetracycline in manure depending on various environmental conditions. They found the breakdown slowed with increased saturation of the manure and concluded this was a result of decreased oxygen levels. This research helps producers understand the effects of oxytetracycline in animal feed on the environment, bacteria, and antimicrobial resistance.
The drug’s withdrawal period and its half-life depend on the type of animal.
| Animal | Half-life | Withdrawal Period |
| Dogs | 4-6 hours | No data |
| Cattle and horses | 4,3-9,7 hours | 6 days |
| Pigs | 6.7 hours | No data |
| Sheep and goats | 3.6 hours | 14 days |
Important Safety information
The spray should be kept away from open flame. When spraying, do not twist up and down. The drug should not be inhaled. Avoid contact with eyes and mucous membranes. Not for human use.
Discontinue use for at least 28 days in cattle and 28 days in swine before slaughter to permit elimination of the drug from edible tissues. Milk taken from animals during treatment and 96 hours after the last treatment must not be used for food.
A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal.
Exceeding the highest recommended dosage level of drug per lb of body weight per day and administering more than the recommended number of treatments may result in antibiotic residues beyond the withdrawal period.
Consult your veterinarian before administering this product to determine the proper treatment for an adverse reaction. At the first sign of any adverse reaction, discontinue the use of the product and seek the advice of your veterinarian. Some of the reactions may be connected either with anaphylaxis (an allergic reaction) or with the cardiovascular collapse of an unknown cause.
As with all antibiotic preparations, the use of this drug may result in the overgrowth of nonsusceptible organisms, including fungi. In addition, a lack of response by the treated animal, or the development of new signs, may suggest an overgrowth of nonsusceptible organisms. If any of these conditions occur, consult your veterinarian.
Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving oxytetracycline in conjunction with penicillin.
While generally safe and effective when prescribed by a veterinarian, oxytetracycline can cause side effects in some animals.
Oxytetracycline should not be used in animals with known hypersensitivity or allergy to the drug. In addition, it should be used with caution in pets with liver or kidney (renal) disease.
Oxytetracycline may decrease appetite and cause stomach, kidney, and intestine problems at high doses.
Oxytetracycline may interact with other medications. Consult your veterinarian to determine if your pet’s other drugs could interact with oxytetracycline. Such drugs include oral antacids.
Oxytetracycline and other tetracyclines can discolor the teeth in young animals. Therefore, do not administer until the adult teeth have fully erupted.
Oxytetracycline should be avoided in pregnant animals as liver problems may occur.
| Oxytetracycline (OTC) Concentrations in Milk Samples Determined by HPLC Method | |||||
| Periods after the first OTC administration (Hours) | Concentrations of OTC in Milk (mg/kg) | Positive Samples/ Total Number | Percentages | ||
| Range | Mean | Standard Deviation | |||
| 0 | Not detected | Not detected | Not detected | 0/6 | 0 |
| 24 | 7.37-185.92 | 59.13 | 70.63 | 6/6 | 100 |
| 48 | 17.26-105.47 | 62.56 | 33.45 | 6/6 | 100 |
| 72 | 35.09-92.45 | 57.70 | 22.67 | 6/6 | 100 |
| 96 | 80.78-351.58 | 195.68 | 94.29 | 6/6 | 100 |
| 120 | 0.72-7.11 | 2.78 | 2.46 | 6/6 | 100 |
| 144 | Not detected-1.58 | 0.39 | 0.60 | 4/6 | 67 |
| 168 | Not detected-0.18 | 0.03 | 0.07 | 2/6 | 33 |
| 192 | Not detected | Not detected | Not detected | 0/6 | 0 |
Care of Sick Animals
The use of antibiotics in the management of diseases is based on an accurate diagnosis and an adequate course of treatment. When properly used to treat diseases caused by oxytetracycline-susceptible organisms, most animals treated with oxytetracycline show a noticeable improvement within 24-48 hours. It is recommended that a veterinarian carry out the diagnosis and treatment of animal diseases. Since many diseases look alike but require different types of treatment, the use of professional veterinary and laboratory services can reduce treatment time, costs, and needless losses. Suitable housing, sanitation, and nutrition are important in maintaining healthy animals and are essential in treating animals.
Veterinary Prescription
Oxytetracycline is a prescription drug that can only be obtained from a veterinarian or by prescription.
To ensure the bacteria causing an infection are susceptible to oxytetracycline, a tissue sample is usually taken, for example, a swab from the infected area or a urine or blood sample.
Prior diagnosis of infection before treatment with antibiotics is recommended in food animals, and using these drugs responsibly in animals for medical reasons is essential for the general well-being of animals. This also prevents certain zoonotic infections from being transferred from animal origin to humans.
Side Effects
From the moment of the first use of Ashoxy 200 ml spray and before the first signs of exposure to the animal’s body, it may take 5 to 10 days. Therefore, carefully check the treatment flow to avoid side effects.
In the course of the clinical evaluation of oxytetracycline, an impressive proportion of patients receiving that antibiotic in tablets were noted as having diarrhea, with or without upper gastrointestinal symptoms; in many of these patients, hemolytic coagulase-positive strains of Staphylococcus aureus were found in pure culture or as the predominant organisms in the watery feces. Some of the staphylococcic diarrheas were quite severe and may have caused or contributed to the death of a few patients.
The Ashoxy may cause effects such as discoloration of the animals’ teeth. Sometimes, after taking that drug, the teeth turned brown. Also, a high dosage of the drug can provoke a decrease in the growth of the animal. Sometimes users noted that after taking Ashoxy, the animals’ bones were restored more slowly.
The antibiotic oxytetracycline can sometimes cause nausea, lack of appetite, vomiting, and diarrhea.
Reports of other adverse reactions associated with oxytetracycline administration include restlessness, ataxia, trembling, swelling of eyelids, ears, muzzle, anus and vulva (or scrotum and sheath in males), respiratory abnormalities (labored breathing), frothing at the mouth, collapse and possibly death. Some of these reactions may be attributed to anaphylaxis (an allergic reaction) or cardiovascular collapse of unknown cause. For a comprehensive list of all possible effects, consult a veterinary physician. If any symptom persists or worsens, or you notice any other symptom, please seek veterinary medical treatment immediately.
Adverse reactions and complications are generally not observed when Ashoxy is used following these instructions.
Ashoxy does not exclude other medications for specific and symptomatic therapy.
Overdose
No symptoms of overdose have been established.
The characteristics of the action on the first application and cancellation of Ashoxy are not disclosed. However, in case of allergic reactions, the use of the drug is discontinued, and antihistamines or other symptomatic treatments are prescribed.
Storage
Ashoxy is stored in the manufacturer’s secure packaging, in a dry place, protected from direct light, separated from food, and feed at a temperature of 15°C to 25°C. The shelf life of the drug, if the storage conditions are met, is two years from the date of production.
After stopping the first dose, the drug must be used within 28 days. And do not forget to dispose of the bottle safely after using the medicinal product.
Ashoxy Advantage
Among the key advantages are low Ashoxy price in 2022 and effectiveness. Other benefits include:
- an optimal set of ingredients;
- high level of safety;
- low toxicity;
- fast therapeutic effect;
- low probability of side effects;
- treats a wide range of diseases, including pinkeye, bovine respiratory disease, foot rot, and leptospirosis;
- effective on various types of cattle.
Drugs Similar to Ashoxy
This drug is registered for use in humans and animals.
Human formulations: Terramycin, Terramycin 200 (Pfizer), Uri-Tet (American Urologicals).
Veterinary formulations: Terramycin (Pfizer), Liquamycin (Pfizer).
Some other names are Oxytetracycline Hydrochloride, Oxyject, Oxytet 1000 SP, and OXY WS.
FAQs
Where Can I purchase Ashoxy Spray or Tablets?
You can buy Ashoxy Spray and Tablets online with a product quality guarantee. Since it became popular in 2022, you will easily find it in many online pharmacy stores.
What Does Oxytetracycline Treat in Animals?
Oxytetracycline is used in different animals to treat bacterial infections, including respiratory infections of the sinuses, wound infections, pneumonia, infections of the oral cavity, and infections of the blood cells.
How Long Does Oxytetracycline Take to Work?
It depends on the condition you treat. You should notice the improvement in 2-3 days of using the antibiotic. Your vet prescribes the dosage and duration. Follow the directions even if your animal feels better.
Is Oxytetracycline Safe for Pregnant Cows?
Suitable for use in pregnant and lactating animals but not in ewes producing milk for human consumption. None known.
Can You Overdose on Oxytetracycline?
Per the patient leaflet that comes with this medicine, there are no specific oxytetracycline overdose problems or symptoms.
Can I Buy Oxytetracycline Over the Counter?
Oxytetracycline is a prescription-only antibiotic used to treat different bacterial infections.
Is Oxytetracycline the Same as Doxycycline?
Doxycycline is a broad-spectrum antibiotic synthetically derived from oxytetracycline. This drug is a second-generation tetracycline, exhibiting lesser toxicity than first-generation tetracyclines.
Does Ashoxy Expire?
Yes, it does. Like all medications, Ashoxy has its expiration date. Check it on the package. Do not give it to the animal if the medicine is expired since it can be toxic.
Can Oxytetracycline Cause Abortion in Cow?
One research has shown that thirty-eight (38%) veterinarians indicated oxytetracycline induces abortion in dairy cows. However, 62% did not ascertain this. Another research has shown that fifty-two (52%) of veterinarians responded that oxytetracycline induces abortion in dairy cows. However, 48% had a negative response. Therefore, you need to consult your veterinarian to get more information.
What’s the Difference Between Oxytetracycline and Tetracycline?
Oxytetracycline is a broad-spectrum tetracycline antibiotic, the second of the group to be discovered. Oxytetracycline works by interfering with the ability of bacteria to produce essential proteins. Without these proteins, the bacteria cannot grow, multiply and increase in numbers. Tetracycline is the first-generation antibiotic.
What Colour is Oxytetracycline?
Tetracycline HCl is “a yellow, odorless, hygroscopic, crystalline powder.
Does Oxytetracycline Affect the Liver?
Several lines of evidence show that oxytetracycline produces severe microvesicular steatosis of the liver, and it has been reported that an excessive dose of oxytetracycline produces hepatic damage.
Is It Safe to Use Antibiotics in Livestock and Pets?
Antibiotic use in food animals can help treat, control, and prevent bacterial diseases in animals. However, antibiotics should only be used when necessary to slow the spread of antibiotic resistance.
Can Oxytetracycline Treat Coccidiosis?
Yes, it can. By 2022, the researchers have proved that oxytetracycline can effectively treat coccidiosis in small ruminants.
What Is the Toxicity of Tetracycline?
The toxicity of tetracycline antibiotics was proven, by many studies, in which induction of fatty liver, acute pancreatitis, and severe hepato-nephrotoxicity contribute to the development and severity of microvesicular steatosis, increase in the triglycerides, AST, ALT, bilirubin, urea, creatinine and gamma-globulin levels.
Is Oxytetracycline Safe for Dogs?
While generally safe and effective when prescribed by a veterinarian, oxytetracycline can cause side effects in some animals. Oxytetracycline should not be used in animals with known hypersensitivity or allergy to the drug. In addition, it should be used cautiously in pets with liver or kidney (renal) disease.
Oxytetracycline is used in dogs and cats to treat bacterial infections, including respiratory infections of the sinuses, wound infections, pneumonia, and infections of the oral cavity and the blood cells.
What Is the Withdrawal Period for Oxytetracycline in Cattle?
Withdrawal periods of 5–20 days are recommended before slaughter, depending on the species and the nature of the food products.
Does Milk Contain Oxytetracycline?
The analysis data showed that oxytetracycline persisted in milk for as long as two days after both treatments at the concentration of 0.03 mg/kg versus 0.02 mg/kg, respectively. Furthermore, no residues were detected in the milk of any animal from the 4th day of the cessation of the therapy as detected by the HPLC method.
What Is the Best Medicine for Pink Eye in Cattle?
A widespread pinkeye epidemic in a herd can be effectively treated with oxytetracycline, followed by feeding the antibiotic in the ration. When the disease is less prevalent, injecting affected animals with penicillin or oxytetracycline is effective.
What Is the Best Antibiotic for Foot Rot in Cattle?
Approved antibiotics for treating foot rot include Naxcel, Nuflor, Liquamycin LA-200, and other long-acting oxytetracycline, Sulmet, and other sulfamethazine boluses, sulfadimethoxine oral solution or powder, and tetracycline powder.

































