Table of Contents
The Vetmedin (pimobendan) tablets were created for dogs with congestive heart failure (CHF) caused by two types of heart disease: Mitral Valve Disease (MVD and/or tricuspid regurgitation) and Dilated Cardiomyopathy (DCM). This medication was the only FDA-approved inodilator by 2022 that helps extend life expectancy and improves the quality of life in dogs. Vetmedin Tablets can also treat dogs with asymptomatic MVD and Dobermans with asymptomatic DCM.
Dogs with CHF have weakened circulatory systems; pimobendan helps them engage in the daily activities they enjoy. But unfortunately, the older dogs get, the more susceptible they are to heart disease and heart failure.
Why Is Maintaining Cardiac Output So Important?
For the body to function correctly, the heart needs to pump blood at a sufficient rate to maintain an adequate and continuous supply of oxygen and other nutrients to the brain and other vital organs. Cardiac output is the term that describes the amount of blood the heart pumps each minute. Doctors think about cardiac output in the following equation: Cardiac output = stroke volume × heart rate.
The stroke volume is the amount of blood the heart pumps each time it beats, and the heart rate is the number of times the heart beats per minute. That is why it is vital to maintain cardiac output with the help of Vetmedin.
Breeds at risk for AVVI (atrioventricular valvular insufficiency) are small to medium-sized dogs such as:
- Chihuahuas;
- Toy poodles;
- Boston terrier;
- Pomeranians.
The middle-aged large and giant breeds are more prone to DCM:
- Newfoundland;
- St. Bernard;
- Boxer;
- Great Dane;
- Doberman pinscher.
What Is Vetmedin?
It is a benzimidazole-pyridazine derivative, a non-sympathomimetic, non-glycoside inotropic drug with vasodilatative properties. The chemical name of pimobendan is 4,5-dihydro-6-[2-(4-methoxyphenyl)-1H-benzimidazole5-yl]-5-methyl-3(2H)-pyridazinone.
By 2022 this medication was available in oblong, half-scored chewable tablets (1.25 mg, 2.5 mg, 5 mg, or 10 mg). The tablets are brownish in color and oval in shape and are scored on both sides so that they can be divided into equal parts. There is no problem quartering the tabs as long as they break relatively cleanly. The drug also comes in capsules (1.25, 2.5, and 5.0 mg), injection solutions (0.75 mg/ml), and liquid form (containing pimobendan 3.5mg/mL), but never as soluble powder. The capsules are hard and are white to orange in color. The form of the medication is prescribed by your vet, considering the pet’s condition.
The positive inotropic effects of medication are mediated through a combination of the following:
- Sensitization of the cardiac contractile apparatus to intracellular calcium. Calcium sensitization results in a positive inotropic effect without increasing myocardial oxygen demand.
- Increased cyclic adenosine monophosphate mediated by phosphodiesterase III (PDEIII) inhibition.
This medication will work by inhibiting the function of a certain enzyme called phosphodiesterase (PDE). Widening blood vessels and, thus, lowering blood pressure allows animals with heart disorders to have a better cardiovascular system. It reduces the work your dog’s heart must do to pump the blood around the body. It also increases the strength of your dog’s heartbeat and cardiac output, improving the pumping action without raising oxygen or energy consumption.
Vasodilation
Balanced vasodilatory effects are mediated through PDEIII inhibition in arterial and venous vascular smooth muscle. Additional endothelial-mediated vasodilation mechanisms may also contribute to this action and may be linked to the medication’s beneficial effect in treating pulmonary hypertension.
Anticoagulation and Other Properties
PDEIII inhibition mediates antithrombotic properties in platelets, leading to reduced platelet aggregation. It has been investigated in other pyridazine-based compounds as well as pimobendan. In a study of healthy dogs, pimobendan mildly inhibited platelet aggregation but at a concentration well above a clinically relevant dose. In addition, some researchers have shown that pimobendan reduced some adverse cytokine concentrations.
Effectiveness
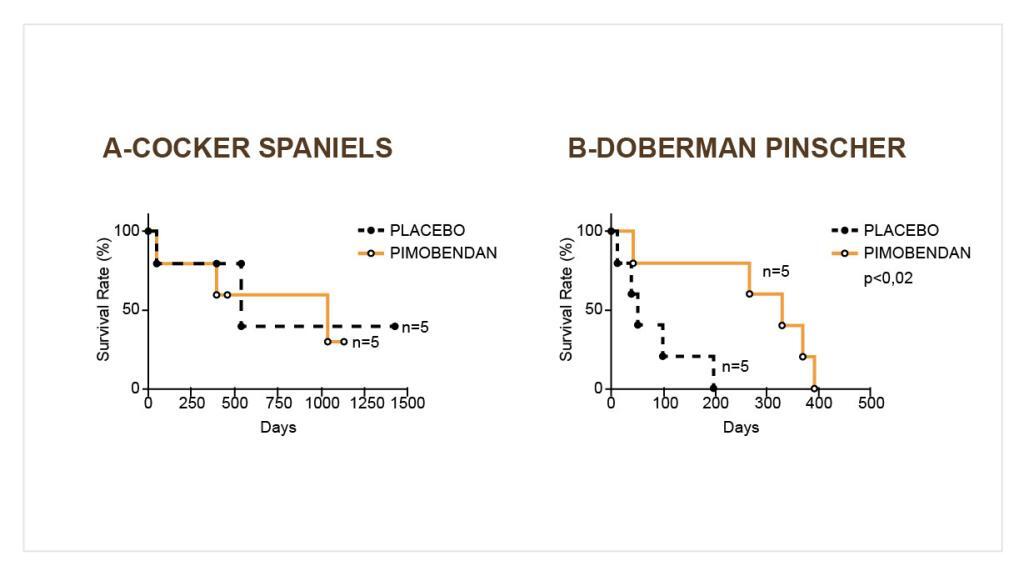
In a 56-day field study, 355 dogs with modified NYHA Class II, III, or IV CHF due to AVVI or DCM were randomly assigned to either the active control (enalapril maleate) or the Vetmedin (pimobendan) treatment group. Of the 355 dogs, 52% were male, and 48% were female; 72% were diagnosed with AVVI, and 28% were diagnosed with DCM; 34% had Class II, 47% had Class III, and 19% had Class IV CHF—dogs in age and weight from 1 to 17 years and 3.3 to 191 lb, respectively. The most common breeds were mixed breeds, Doberman Pinscher, Cocker Spaniel, Miniature/Toy Poodle, Maltese, Chihuahua, Miniature Schnauzer, Dachshund, and Cavalier King Charles Spaniel.
The 180 dogs (130 AVVI, 50 DCM) in the active control group received enalapril maleate (0.5 mg/kg once or twice daily), and all except two received furosemide. Per protocol, all dogs with DCM in the active control group received digoxin. The 175 dogs (126 AVVI, 49 DCM) in the Vetmedin group received pimobendan (0.5 mg/kg/day divided into two portions that were not necessarily equal, and the portions were administered approximately 12 hours apart), and all but 4 received furosemide. Digoxin was optional for treating supraventricular tachyarrhythmia in either treatment group, as was the addition of a β-adrenergic blocker if digoxin was ineffective in controlling heart rate. After initial treatment at the clinic on Day 1, dog owners were to administer the assigned product and concurrent medications for up to 56±4 days. The determination of effectiveness (treatment success) for each case was based on improvement in at least two of the three following primary variables:
- modified NYHA classification;
- pulmonary edema score by a masked veterinary radiologist;
- the investigator’s overall clinical effectiveness score (based on physical examination, radiography, electrocardiography, and clinical pathology).
Attitude, pleural effusion, coughing, activity level, furosemide dosage change, cardiac size, body weight, survival, and owner observations were secondary evaluations contributing information supportive to product effectiveness and safety. Based on protocol compliance and individual case integrity, 265 cases (134 Vetmedin, 131 active control) were evaluated for treatment success on Day 29. See Table 1 for effectiveness results.
Table: Results for the 56-Day Field Research
| Vetmedin (Pimobendan) | Active Control Group | |
| Success in Treatment (Day 29) | 80.7% n=134 | 76.3% n=131 |
| 88 of 101 dogs with AVVI | 77 of 100 dogs with AVVI | |
| 20 of 33 dogs with DCM | 23 of 31 dogs with DCM | |
| Success in Treatment (Day 56) | 71.1% n=113 | 67.2% n=110 |
| 66 of 85 dogs with AVVI | 56 of 85 dogs with AVVI | |
| 13 of 28 dogs with DCM | 17 of 25 dogs with DCM |
At the end of the 56-day study, dogs in the Vetmedin group were enrolled in an unmasked field study to monitor safety under extended use without restrictions on concurrent medications. As a result, Vetmedin was used safely in dogs concurrently receiving furosemide, enalapril, digoxin, atenolol, nitroglycerin, diltiazem, spironolactone, hydralazine, antiparasitic products (including heartworm prevention), antibiotics (amoxicillin-clavulanate, metronidazole, fluoroquinolones, cephalexin), topical ophthalmic and otic products, theophylline, famotidine, levothyroxine sodium, butorphanol, hydrocodone, diphenhydramine, and metoclopramide, and in dogs on sodium-restricted diets.
Check the effectiveness and safety of pimobendan in heart failure in dogs caused by mitral valve endocarditis in the picture below.
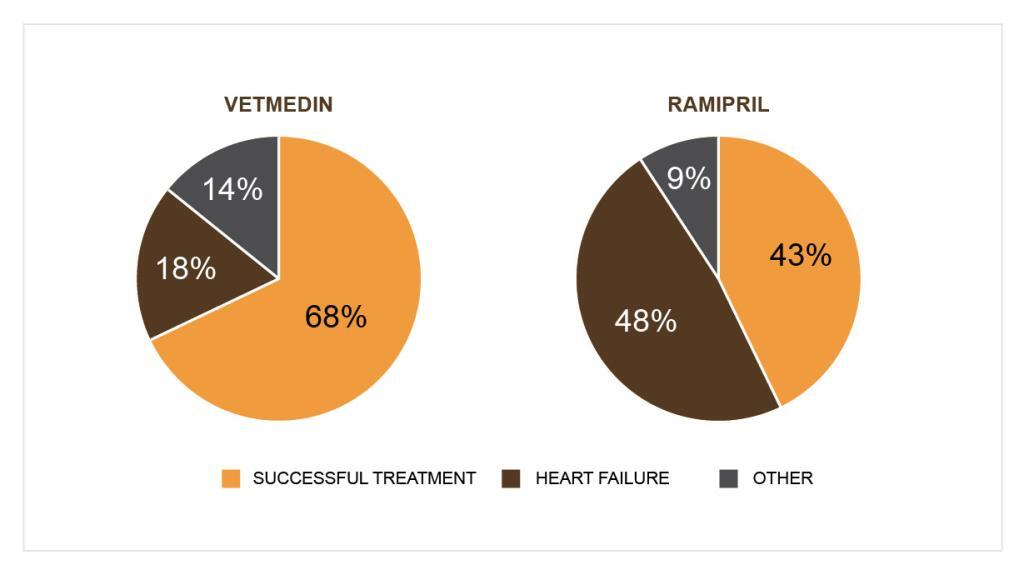
Additional therapy with Vetmedin resulted in improved clinical symptoms and decreased mortality compared with standard treatment.
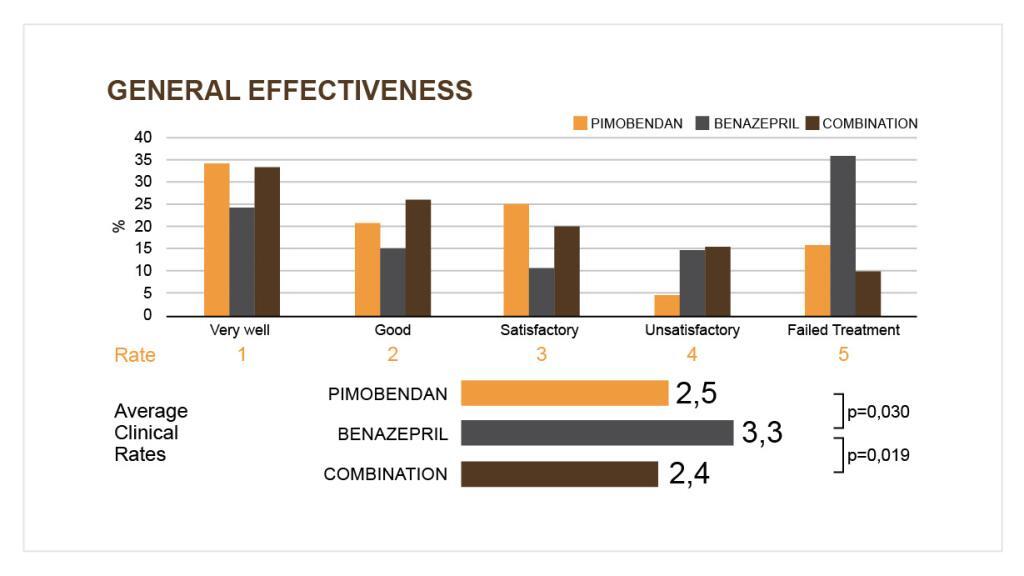
Treatment with Vetmedin in dogs with congestive heart failure resulted in a statistically significant improvement in clinical symptoms, which could also lead to an improvement in the quality of life of the animals. However, the combination of Vetmedin + benazepril did not lead to better clinical results than Vetmedin monotherapy.
Ingredients
The positive effects are achieved due to the ingredients of the medicine. The main component of the medication is pimobendan, a benzimidazole-pyridazine derivative. It is a non-glycoside and non-sympathomimetic inotropic substance that has vasodilatative properties.
Uses
It is prescribed for dogs with congestive heart failure in different stages with or without structural abnormality with or without clinical signs of the animal’s heart failure. In general, there are two common uses:
- to treat congestive heart failure that comes from valvular insufficiency or dilated cardiomyopathy;
- to treat Doberman Pinscher dogs diagnosed with clinically asymptomatic dilated cardiomyopathy to delay the onset of sudden death or congestive heart failure.
American College of Veterinary Internal Medicine (ACVIM) issued the following guidelines for CHF treatment:
|
STAGE A |
STAGE В |
STAGE C | STAGE D |
|
Dogs with a high risk of heart disorder |
A murmur (an abnormal heart sound because of abnormal blood flow) is heard, but there are no signs of heart failure. |
Signs of heart failure are visible, and the dog needs treatment | Heart failure that is hard to manage, and the dog doesn’t respond to standard treatment. |
| STAGE B1: The heart is not changed or enlarged on an X-ray | |||
| STAGE B2: The heart is changed or enlarged on an X-ray |
Also, please check the table below to find out the use of the medication in dogs with heart disorders according to the stage of the disease:
| HEART DISEASE | STAGE | VETMEDIN INDICATION |
| Dilated cardiomyopathy | Stage B2 |
|
| Stages C and D | Recommended | |
| Degenerative mitral valve disease | Stage B2 | Recommended in dogs that meet EPIC entry criteria |
| Stages С and D | Recommended | |
| Congenital heart disease with volume overload complicated by congestive heart failure | Consider | |
| Congenital heart disease characterized by obstruction with secondary myocardial failure respond to standard treatment | Consider | |
| Pulmonary hypertension resulting in right-sided CHF refractory to standard treatment | Consider | |
| Pericardial effusion that needs pericardiocentesis and results in ascites | Contraindicated | |
Diagnosis based on echocardiography.
Consult your veterinarian before starting the medication.
Other Indications
- Congenital heart disease characterized by obstruction (e.g., subaortic stenosis, pulmonic stenosis) with secondary myocardial failure in a patient with symptoms refractory to standard therapy. We strongly recommend consultation with a cardiologist before taking pimobendan for these cases.
- Congenital heart disease with volume overload (e.g., ventricular septal defect, patent ductus arteriosus, atrial septal defect, mitral or tricuspid valve dysplasia) complicated by CHF.
- Pulmonary hypertension unrelated to DMVD, resulting in right-sided CHF (ascites) refractory to standard therapy (i.e., cor pulmonale, advanced heartworm disease, pulmonary thromboembolism). For these patients, pimobendan can be added to other medicines; however, we recommend first consulting a vet.
Dosage Sizes
The dosage depends on the dog’s weight. In addition, pet owners can buy medicine in chewable pills, capsules, injections, and oral liquid.
For dogs, the labeled dosage recommendation for pimobendan is 0.23 mg/lb (0.5 mg/kg) body weight, using a suitable combination of whole or half tablets. The total daily dose can be administered in 2 unequal portions using whole or half tablets.
For initial use, especially if a more rapid onset of action is desired, the tablets should be administered on an empty stomach; however, they can be taken with food for more chronic use. The oral preparation is rapidly absorbed; the peak effect occurs within 2 to 4 hours in dogs and 0.9 hours in cats.
Dosage Instructions
The right way to choose the proper dosage is to consult a professional vet. But overall, the daily dose ranges between 0.2 mg and 0.6 mg of pimobendan per 1 kg of body weight. Moreover, the preferable daily dosage is 0.5 mg per 1kg of body weight. This dose must be divided into two intakes of 0.25 mg per 1 kg. Give one half in the morning and wait 12 hours before taking the other. For more detailed information, please check the table below.
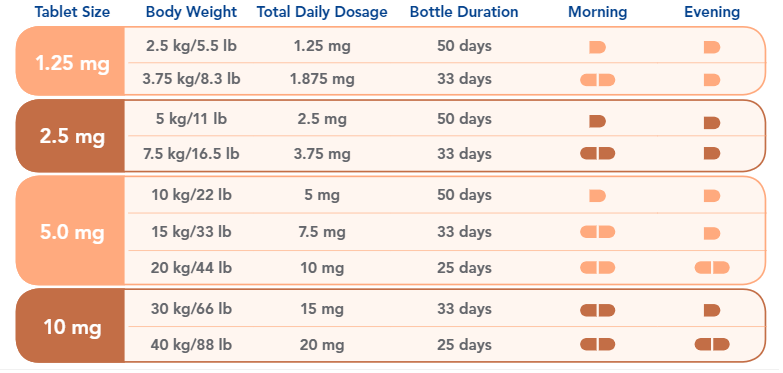
Please note: These examples in the dosage table do not necessarily depict the only way to provide the correct dose.
Other combinations of whole and half tablets may also be appropriate.
Helpful Tip: If your dog refuses to take the medication, try to hide it in a tasty treat or pill pocket.
Withdrawal Time
The withdrawal time varies according to the dosage for dogs concerning their weight. Metabolically it gets out of the system in about 2 hours. Since the active ingredient is metabolized mainly via the liver, it should not be used in dogs with severe impairment of liver function.
Important Safety Information for Dog Owners
If you think of getting the medication for your dog, remember that the dog should be older than six months, not pregnant or breastfeeding, without cardiomyopathy, aortic stenosis, or conditions that include any risks due to increased blood pressure. Due to the ingredients in the tablets, the dosage prescription should be strictly followed not to overcome the acceptable level of toxicity in the body. These tablets must be used only when the dogs have clear evidence of CHF, and if you’ve purchased the medication, read its description. Here is some more important information:
- Blood glucose should be tested regularly during treatment in dogs with diabetes mellitus.
- For use in the “preclinical stage” of dilated cardiomyopathy (asymptomatic with an increase in left ventricular end-systolic and end-diastolic diameter), a diagnosis should be made through a comprehensive cardiac examination (incl. echocardiographic examination and possibly Holter monitoring).
- For use in the preclinical stage of myxomatous mitral valve disease (stage B2, according to ACVIM consensus: asymptomatic with mitral murmur ≥3/6 and cardiomegaly due to myxomatous mitral valve disease), a diagnosis should be made through a comprehensive physical and cardiac examination which should include echocardiography or radiography where appropriate. (See also ‘Further Information’ section).
- Monitoring of cardiac function and morphology is recommended in animals treated with pimobendan.
- The product’s safety has not been assessed in nursing or pregnant dogs. Use only according to the benefit/risk assessment by the responsible veterinarian.
- In prolonged exposure (six months) of healthy beagle dogs at 3 and 5 times the recommended dose, mitral valve thickening and left ventricular hypertrophy were observed in some dogs. These changes are of pharmacodynamic origin.
- No interaction between pimobendan and the cardiac glycoside strophanthin was observed in pharmacological studies. The pimobendan-induced increase in cardiac contractility is attenuated by the calcium antagonists verapamil and diltiazem and by the β-antagonist propranolol.
- Laboratory studies on rats and rabbits have not produced evidence of teratogenic or foetotoxic effects. However, these studies have shown evidence of maternotoxic and embryotoxic effects at high doses and that pimobendan is excreted into milk.
In case of accidental ingestion, seek medical advice immediately and show the package leaflet or the label to the veterinarian—wash hands after use.
Advice for doctors: accidental ingestion, especially by a child, may lead to orthostatic hypotension, tachycardia, headaches, and face flushing.
Palatability
In a laboratory study, the palatability of the medication has evaluated in 20 adult female Beagle dogs offered doses twice daily for 14 days. Ninety percent (18 of 20 dogs) voluntarily consumed over 70% of the 28 tablets. Including two dogs that consumed only 4 and 7% of the tablets offered, the average voluntary consumption was 84.2%.
Veterinary Prescription
By 2022 veterinary prescription of Vetmedin for dogs is necessary since there should be evidence of the clinical heart condition of the dog. However, there are several cases when the animal shouldn’t be getting this medication, including healthy dogs that can develop abnormal pathologies.
Contraindications and Warnings
Medication is safe and well tolerated. Reported adverse effects are relatively rare and are typically limited to gastrointestinal upset associated with chewable tablets. However, a significant overdose has reportedly resulted in pimobendan toxicity, manifested as severe tachycardia and mild changes in blood pressure.
Before anyone decides to buy the pills, it is necessary to know that they can’t be given to dogs which:
- are younger than six months old;
- are breastfeeding, pregnant, or giving birth;
- are used for breeding;
- have diseases in which an improvement in cardiac output cannot be achieved for functional or anatomical reasons (e.g., aortic stenosis);
- have hypertrophic cardiomyopathy;
- have severe impairment of liver function;
- have any conditions when heart pressure isn’t recommended to be increased.
Tell your veterinarian if your dog takes verapamil (Isoptin, Calan, Verelan), propranolol (Inderal), theophylline (Theo-Dur), and pentoxifylline (Trental). Talk to your veterinarian or pharmacist before giving any prescription or over-the-counter medicines, including vitamins and minerals.
Tell your veterinarian about your dog’s congenital heart defects, diabetes, or other serious metabolic diseases. Also, tell your veterinarian if your dog is pregnant or lactating.
The product has not been tested in asymptomatic myxomatous mitral valve disease in dogs with significant supraventricular and/or ventricular tachyarrhythmia.
The product has not been tested in cases of asymptomatic DCM in Dobermans with atrial fibrillation or sustained ventricular tachycardia.
Vetmedin should be used cautiously in patients with an outflow tract obstruction, which includes the following:
- pulmonic stenosis;
- subaortic stenosis;
- asymmetric septal hypertrophy;
- the systolic anterior motion of the mitral valve.
These tablets should be used only to help dogs with a medical condition of congestive heart failure.
Suppose pimobendan is given to a patient with outflow tract obstruction or low systemic blood pressure to ensure it is well tolerated. In that case, you should reassess blood pressure and heart rate approximately 1 to 2 hours after the first dose of pimobendan is given.
Side Effects
The frequency of adverse reactions is defined using the following convention:
– very common (more than 1 in 10 animals treated displaying adverse reactions);
– common (more than one but less than ten animals in 100 animals treated);
– uncommon (more than one but less than ten animals in 1,000 animals treated);
– rare (more than one but less than ten animals in 10,000 animals treated);
– very rare (less than one animal in 10,000 animals treated, including isolated reports).
Side effects in dogs can happen due to overdose. They may include vomiting, low appetite, lethargy, hyperactivity, shortness of breath, erythema, constipation, diarrhea, rash, bleeding, weakness, anemia, drooling, ataxia (lack of muscle control and coordination), excited behavior, low blood pressure, or increased heart rate. Contact your local veterinary clinic if you notice any side effects.
- A slight chronotropic effect (rise in heart rate) and vomiting can occur in rare cases. However, these effects are dose-dependent and can be avoided by reducing the dose.
- In rare cases, transient diarrhea, anorexia, or lethargy have been observed.
- In rare cases, mitral valve regurgitation has been observed during chronic pimobendan treatment in dogs with mitral valve disease.
- Although a relationship with pimobendan has not been established, signs of effects on primary hemostasis (petechiae on mucous membranes, subcutaneous hemorrhages) may be observed during treatment in sporadic cases. These signs disappear when the treatment is withdrawn.
Severe side effects that should be immediately reported to the vet:
- an allergic reaction (difficulty breathing; swelling of the lips, tongue, or face; hives);
- scratching;
- sudden onset of diarrhea, vomiting;
- shock, seizures, pale gums
- coma.
Contact your vet and continue treatment if you notice the following conditions:
- incoordination;
- fainting;
- accumulation of fluid in the lungs or abdomen;
- cough.
Clinical findings/adverse reactions were recorded in a 56-day field study of dogs with congestive heart failure (CHF) due to AVVI (256 dogs) or DCM (99 dogs). Dogs were treated with either Vetmedin (175 dogs) or the active control enalapril maleate (180 dogs). Dogs in both treatment groups received additional background cardiac therapy. The Vetmedin group had the following prevalence (percent of dogs with at least one occurrence) of common adverse reactions/new clinical findings (not present in a dog before beginning study treatments): poor appetite (38%), lethargy (33%), diarrhea (30%), dyspnea (29%), azotemia (14%), weakness and ataxia (13%), pleural effusion (10%), syncope (9%), cough (7%), sudden death (6%), ascites (6%), and a heart murmur (3%). Prevalence was similar in the active control group. However, the prevalence of renal failure was higher in the active control group (4%) compared to the Vetmedin group (1%).
Table: CHF Death and New Arrhythmias in the 56-Day Field Research
| Vetmedin Group | Active Control Group | |
| Dogs died from CHF | 14.3%, n=175 | 14.4%, n=180 |
| 9 of 126 dogs with AVVI | 16 of 130 dogs with AVVI | |
| 16 of49 dogs with DCM | 10 of 50 dogs with DCM | |
| Dogs with new arrhythmias | 39.4%, n=175 | 45.0%, n=180 |
| 45 of 126 dogs with AVVI | 59 of 130 dogs with AVVI | |
| 24 of 49 dogs with DCM | 22 of 50 dogs with DCM |
Overdose
If the dog has an overdose, you can notice such symptoms as a positive chronotropic effect, vomiting, convulsions, loss of appetite, drowsiness, labored breathing, weakness, incoordination, fainting, accumulation of fluid in the lungs or abdomen, cough, collapse, diarrhea, apathy, ataxia, heart murmurs or hypotension, fast heart beating, and significant decreasing of blood pressure. It is because the medication excess gives an increased toxicity level. In this situation, the dosage should be reduced, and appropriate symptomatic treatment should be initiated. That’s why it is crucial to contact the nearest animal clinic as soon as possible if any of these symptoms occur.
Poisoning
There are two types of pimobendan poisoning: acute (taking a large dose causing immediate toxicosis) or chronic (taking small amounts regularly, causing a gradual toxicosis).
Causes of Poisoning in Dogs
- accidental overdose by the owner;
- dog ingested medication;
- giving the drug to the wrong dog.
Signs of Toxicity
Your dog may not show any signs of toxicity immediately, and some mild symptoms, such as stomach upset and weakness, may be considered a virus or anxiety. Anytime you think your dog may have ingested a medication, you must call your veterinarian or get your dog to a pet hospital or clinic. Some of the most commonly reported symptoms are:
- cough;
- diarrhea;
- allergic reaction (breathing difficulty, hives);
- collapse;
- gagging;
- irregular pulse;
- dark sticky stool (melena);
- dehydration;
- trembling;
- fainting (syncope);
- vomiting;
- increased respiratory rate;
- lethargy;
- depression
- loss of bodily movements (ataxia);
- elevated heart rate
- restlessness;
- fluid accumulation in the lungs and/or abdomen (ascites)
- shaking;
- heart murmur;
- sudden death;
- increased thirst and urination;
- weakness;
- itchy skin (pruritus);
- loss of appetite;
- low blood pressure;
- seizures;
- staggering;
- urinary accidents;
- weak pulse;
- weight loss.
Diagnosis of Poisoning in Dogs
A vet will do a complete body examination, including vision and hearing check, reflexes, abdominal palpation, oxygen level, heart rate, body temperature, and blood pressure.
Some tests, such as complete blood count (CBC), serum electrolyte levels, urinalysis, protein and glucose, and fecal examination, will also be performed.
Chest and abdominal images through radiographs (x-rays) may be done to examine the heart and lungs. If the veterinarian suspects damage, CT scans, ultrasound, and possibly an MRI may be used to get a better view. An electrocardiogram (ECG) can measure the heart’s electrical and muscular activity.
Treatment of Poisoning in Dogs
The treatment for acute poisoning is to induce vomiting and administer activated charcoal to bind with the medication to eliminate it more safely.
In chronic poisoning cases, the toxicity damage has already been done, so the treatment will depend on which organs are affected.
Animal Safety
In a laboratory study, chewable tablets were administered to 6 healthy Beagles per treatment group at 0 (control), 1, 3, and 5 times the recommended dosage for six months. See Table 3 for cardiac pathology results. The cardiac pathology/histopathology noted in the 3X and 5X dose groups are typical of positive inotropic and vasodilator drug toxicity in normal dog hearts and is associated with exaggerated hemodynamic responses to these drugs. None of the dogs developed signs of heart failure, and there was no mortality.
Storage
The tablets are flavored, so they must be stored out of animals’ reach to avoid accidental ingestion.
Recommended storage condition is to keep it in a space with temperatures ranging from 59°F up to 86°F (15 to 30°C).
Benefits of Using
The medication helps dogs suffering from congestive heart disease and makes your pet’s life more comfortable, relieving the symptoms of heart failure. In addition, it increases the ability of the heart to pump blood. Besides, it comes in delicious beef-flavored chewable tablets.
How long does Vetmedin prolong life? If you follow the prescribed dosage, you can expect the animal’s survival to increase more than two times. In addition, its side effects are minor and don’t put vital organs in critical danger.
Drugs Similar to Vetmedin
- Vasotec
- Lanoxin
- Zestril
There is no FDA-approved alternative to this medicine. Capsules, oral liquids, injections, and chewable tablets contain the same active ingredient, pimobendan.
How can i get Vetmedin?
You can buy Vetmedim online.
Delivery
USA, Australia, Canada, Europe, and etc.
Frequently Asked Questions
Does My Dog Need Vetmedin?
It is a critical medication for treating dogs with congestive heart failure due to valvular insufficiency or dilated cardiomyopathy. Unfortunately, there is no FDA-approved alternative to it. So if a vet prescribes it to your dog, your pet must take it.t
What Does This Medicine Do For My Dog?
It widens the blood vessels, so the medication decreases pressure on the animal’s heart and helps the cardio system work better.
Where Can I Buy It?
The tablet supply can be purchased in online shops at an affordable price. You can find the best price for Vetmedin if you get it in a larger set of tablets to save money. The price also depends on the amount of pimobendan in it.
Why is Vetmedin So Hard to Get Sometimes?
It is a prescription drug for pets with heart issues. Sometimes it was in short supply because of production issues related to the COVID-19 pandemic.
Can I Buy Vetmedin Over the Counter?
It differs from other commonly used heart drugs because it helps the heart pump more efficiently; that’s why it requires a prescription from your veterinarian.
Does It Come in Liquid?
Yes, it comes in oral liquid. It is an oil-based medication. It is also available in capsules, chewable pills, and injections (in some countries). The active ingredient in all forms of medicine is pimobendan.
Is Compounded Pimobendan the Same as Vetmedin?
Capsules, liquids, injections, and chewable tablets contain the same active ingredient, pimobendan. Your vet should recommend you the form of the medicine.
What Is the Difference Between Pimobendan and Vetmedin?
Pimobendan is an active ingredient of the medication with the brand name Vetmedin. So Pimobendan is a generic version of the medicine.
How Long Does It Take for Medicine to Start Working?
Every dog is different, and in heart failure, their response depends on how unwell they are. It takes around a week of consistent tablet intake to see the results. Consult your vet if you don’t notice any improvements within seven days of treatment.
How to Know if Medication Is Working?
You should see an improvement within a week after your dog starts treatment, which may get even more noticeable in the following weeks. In dogs with asymptomatic MVD or DCM, you are less likely to notice any noticeable changes in your dog after starting treatment.
How Can I Monitor If My Dog’s Condition Is Getting Worse?
There are two ways in which you can monitor your dog’s condition:
- Regular check-ups with your vet when they can listen to your dog’s heart and learn about their activity level and demeanor at home.
- By monitoring your dog’s breathing rate and keeping a diary of their day-to-day activities, you can track any changes that may help indicate whether their condition is stable or worsening.
How Long Do Dogs Live on Vetmedin?
Once congestive heart failure develops, the survival time of affected dogs is expected to be between 6 and 14 months. Some dogs, however, can live for nearly three years with the proper treatment regimen.
How Long Will My Dog Require Treatment with This Medicine?
For as long as your dog is responding well to treatment. It is likely to mean for the rest of their life. This medicine may be given alone or with other medication during this time.
Is There a Maximum Duration of Administration?
No, there is no limit on the time it can be administered to a patient.
What Should I Avoid While Giving Vetmedin?
It is not for use in humans. Do not give this medication if your dog is allergic to it. Do not give to animals with hypertrophic cardiomyopathy, aortic stenosis, or any other condition where an increased cardiac output is not recommended. Do not give this medication to dogs under six months or pregnant or lactating animals.
How Can I Get My Dog to Take Pill?
The easiest way to give your dog a pill is to hide the pill in food. It usually works best if you hide it in a special treat, such as a little canned dog food, cooked sweet potato, or a soft dog treat that can be molded around it.
Why Is This Medication Taken On an Empty Stomach?
It is not absorbed very well from the stomach if there is food there. It is recommended to administer 30-60 minutes before feeding, but closer to 60 would be better if you can manage it.
Can I Give My Dog Pimobendan With Food?
The total daily dose can be administered in 2 unequal portions using whole or half tablets. For initial use, especially if a more rapid onset of action is desired, the tablets should be administered on an empty stomach; however, they can be taken with food for more chronic use.
Can I Give My Dog Vetmedin an Hour Early?
Yes, this shouldn’t cause any problems at all. But it is better to give the medicine on an empty stomach, 1 hour before food and 3 hours after.
Can It Be Given 2 Hours After Food?
It is better to give the medicine on an empty stomach, 1 hour before food and 3 hours after.
What Happens If I Miss Giving a Dose?
Give the missed dose as soon as you remember. However, if it is almost time for the next regularly scheduled dose, skip the missed dose and give the next one as directed. Do not give a double dose of the medication.
I Accidentally Gave My Dog a Double Dose of Vetmedin. What Should I Do?
This small overdose will not cause your dog any harm. However, you must watch your dog and give the next regular dose at the correct time. If any side effects like gastrointestinal upset, lethargy, and incoordination occur, contact your vet ASAP.
Can I Give the Medicine Once a Day?
If you run out of pills before your new shipment arrives, giving a 1/2 dose twice daily is recommended. It is the standard dosing frequency for this drug. Hopefully, you will not need to do that for over a day or two.
Does the Medication Slow the Heart Rate?
Yes, it can slow down the heart so the ventricles can fill with more blood. Watch your pet and contact the vet if any problems with heart rate occur.
Does Vetmedin Cause Gas in Dogs?
Usually, dogs tolerate it very well. It is rare for side effects to be observed. Some dogs may experience side effects listed in the article, but there is an individual reaction of your pet, such as gas or others. Please consult your vet.
Can the Medicine Cause Kidney Failure in Dogs?
Sometimes, cardiovascular disease can lead to kidney dysfunction, not medication.
How Long Do Side Effects Last?
It has severe and less serious side effects. Some of them may last for a long time. Contact your vet ifs you notice any side effects in your pet.
Can You Compound Vetmedin?
No, you shouldn’t unless your veterinarian has specifically directed you to do so. Prescriptions for compounded medications are written explicitly for individual animals; using one pet’s compounded medication for another could harm your pet.
Can Vetmedin and Enalapril Be Taken Together?
Yes, they can if your vet prescribes it. Don’t hesitate to contact your vet before giving any medications and vitamins to your pet.
Can Vetmedin and Furosemide Be Taken Together?
Yes, they can if your vet prescribes it.
How is the Medication Withdrawn?
The half-life of pimobendan in the blood is 0.4 hours, and the half-life of its metabolite is two hours. The medicine withdrawal is by excretion in the bile and then feces. Pimobendan is 90–95% bound to plasma proteins in circulation.
What Will Happen If I Stop Giving My Dog Vetmedin?
Giving a reduced dose does carry the potential of less control of your dog’s congestive heart failure, so watch for increased coughing, more rapid breathing, or decreased activity; if these are noted, contact your veterinarian immediately.
How to Wean a Dog Off This Medicine?
Pimobendan is a unique and positive inotropic medication that strengthens the heart muscle, especially with an enlarged heart. It is very dangerous to wean a dog off it without consulting your vet.
Can Vetmedin Kill My Dog?
The tablet’s goal is to prolong dogs’ life expectancy, although an overdose of the medication can be fatal. Sometimes, pimobendan has been known to cause rapid heart rate, vomiting, diarrhea, nervousness, collapse, convulsions, and possibly death if not treated immediately. In addition, if given to a dog without a heart condition, the effects can be life-threatening immediately.
Will I Need to Revisit My Vet?
As with any long-term medication, you should regularly take your dog to your vet. At first, these visits may be more frequent. The interval between visits may increase when your dog has been stabilized on Vetmedin. However, if your dog’s condition deteriorates, you may need to visit your vet more often. Your vet will advise you how frequently they must check your dog based on their condition.
Does Vetmedin Expire?
Yes, it does. All medications have an expiration date, usually one to five years after manufacturing. The date is indicated on the package. Only give it to your dog if it is valid.

































